The science of syrup and traffic jams
Summary
An international group of scientists from the UK, Germany, and Australia has discovered the reason why syrup is so sticky by comparing it with traffic jams. When salts or sugars are added to water, the resulting mixture is very ‘sticky’ and flows with great difficulty or – to use the scientific term – it has a high viscosity. This effect is probably most familiar in mixtures of sugars and water such as in syrup and honey.
A debate has been raging for decades as to the scientific reason why things like honey are so sticky. The consensus had been that sugar and salt molecules in water would bind to the water molecules and thereby alter the structure of water. So-called structure inducers (with the beautiful scientific name ‘kosmotropes’) would make liquid water more ice-like and thus stickier... Unfortunately, work by a Dutch group threw a spanner in the works by suggesting that no such structure-inducing effect exists (DOI: 10.1126/science.1084801) leaving chemists and physicists at a loss how to explain stickiness.
A new study published in the Journal of Chemical Physics puts an end to all the confusion (DOI: 10.1063/1.2906132). The team consists of researchers from the University of Regensburg, Murdoch University, and the University of Strathclyde. Leader of the team, Professor Klaas Wynne explains that ‘the German, Australian, and British teams use complimentary techniques making it possible for the first time to understand the motions of water molecules in solution on time scales of about one-trillionth of a second’ (that is: 1/1,000,000,000,000 seconds).
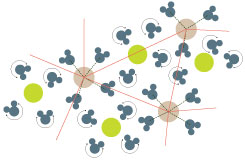
The team studied solutions of metal salts rather than those of sugars because the salt ions are simpler appearing like smooth spheres rather than knobbly molecules. He continues ‘The metal ions we have studied hold on tightly to a few neighbouring water molecules forming tacky spheres that randomly jam together at high enough concentration like cars in a traffic jam.’ His colleague Dr David Turton explains: ‘You can think of a crystal as oranges in a box at the supermarket, neatly arranged in rows and layers. If you instead throw the oranges into the box, they jam into a random jumble – what physicists call – a glass. We find that ions and sugar molecules in water jam like oranges while the water is free to spin in the pockets between the “oranges”.’
This type of jamming has now been seen in the most unlikely collection of situations such as the movement of dunes in the desert, the flow of grains in silos, and – yes – traffic jams. So, next time you are stuck in traffic, think about how you are simply following the laws of physics like sugar in the cup of coffee you had for breakfast that morning.
Notes
The reference to the paper is:
David A. Turton, Johannes Hunger, Glenn Hefter, Richard Buchner, Klaas Wynne, "Glasslike Behavior in Aqueous Electrolyte Solutions", Journal of Chemical Physics, Communication 128, 161102 (2008)
Also of interest is the sister paper:
David A. Turton and Klaas Wynne, "Structural relaxation in the hydrogen-bonding liquids N-methylacetamide and water studied by optical Kerr-effect spectroscopy", Journal of Chemical Physics 128, 154516 (2008)
Images
Click on the thumbnails below for a bigger version and caption.
Further details
We gratefully acknowledge funding for this project from the Engineering and Physical Sciences Research Council (EPSRC).
David A. Turton and Klaas Wynne are in the Biomolecular and Chemical Physics group, in the Department of Physics, SUPA, University of Strathclyde, Glasgow G4 0NG, UK
Johannes Hunger and Richard Buchner are in the Dielectric Relaxation Spectroscopy group, Institut für Physikalische und Theoretische Chemie, Universität Regensburg, D-93040 Regensburg, Germany
Glenn Hefter is in the Chemistry Department, Murdoch University, Murdoch, WA 6150, Australia